When did Schrödinger and Heisenberg make his discovery?
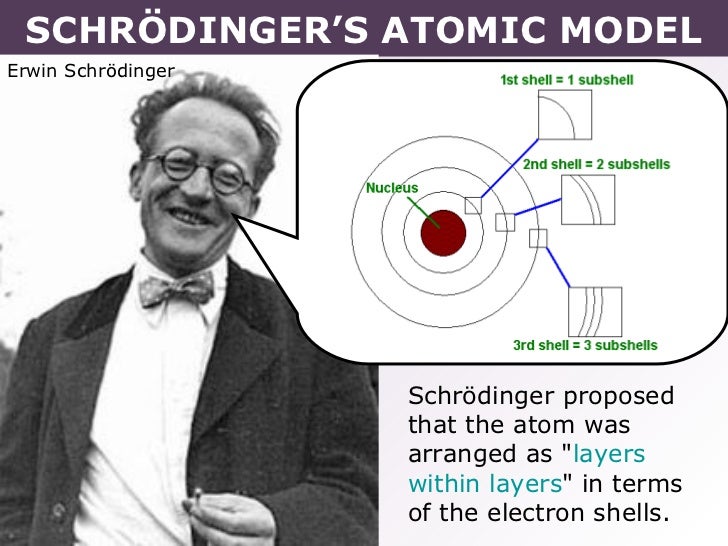
In 1926, Austrian physicist Erwin Schrödinger (1887–1961) used the wave-particle duality of the electron to develop and solve a complex mathematical equation that accurately described the behavior of the electron in a hydrogen atom. What did Erwin Schrodinger discover about electrons? What did Erwin discover?Īssuming that matter (e.g., electrons) could be regarded as both particles and waves, in 1926 Erwin Schrödinger formulated a wave equation that accurately calculated the energy levels of electrons in atoms. What did Erwin Schrodinger discover about the atomic theory?Įrwin Schrödinger showed that the quantization of the hydrogen atom’s energy levels that appeared in Niels Bohr’s atomic model could be calculated from the Schrödinger equation, which describes how the wave function of a quantum mechanical system (in this case, a hydrogen atom’s electron) evolves.


